Full Text Searchable PDF User Manual

Please read these instructions
carefully before use
Compressor
Nebuliser
Instructions
Model: CN02-WD
Engineered for Life
www.clement-clarke.com
Colour: Pantone 2655
Black

Contents
1. Introduction ------------------------------------------------------ Page 2
2. Product Identification -------------------------------------------- Page 2
3. Safety and Regulatory Information ----------------------------- Page 3
4. Operating Your Compressor Nebuliser ------------------------ Page 4
5. Notes on Nebuliser Care ---------------------------------------- Page 5
1. Replacing your consumables
2. Spares and Accessories
3. Optional Accessories
6. Replacement Parts & Optional Accessories ------------------- Page 5
7. Cleaning ---------------------------------------------------------- Page 6
8. Technical Specifications ----------------------------------------- Page 7
9. Guarantee -------------------------------------------------------- Page 8
10. Product Contents ---------------------------------------------- Page 8
11. Symbol Explanations ------------------------------------------ Page 9
12. EMC Declaration ----------------------------------------------- Page 9
1
1. Introduction
Thank you for purchasing the
AirMed 1000 Compressor Nebuliser
. It is a
compact medical device designed to efficiently deliver physician prescribed
medication to the bronchial lung passages. With the correct care and use, it
will provide you with many years of reliable treatment.
This product is developed for the successful treatment of asthma, allergies
and other respiratory disorders. It creates a stream of air that travels through
a clear tube to the nebuliser. When air enters the nebuliser, it will convert the
prescribed medication into aerosol mist for easy inhalation.
Your Compressor Nebuliser should be used under the supervision of a licensed
physician and/or a respiratory therapist. We encourage you to thoroughly read
this handbook to learn about the features of this product. Any use of this
product other than its intended use should always be avoided.
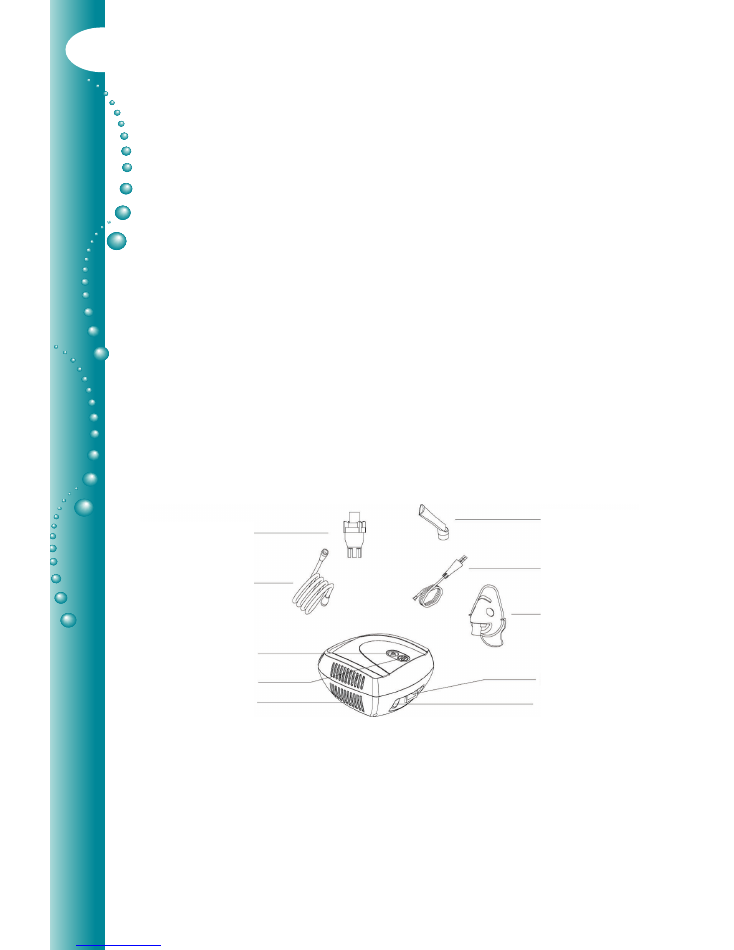
2. Product Identification
1. Nebuliser cup
2. Driveline
Driveline Connector
Filter & Cover
Air Openings
3. Mouthpiece
4. Power Lead
5. Mask
Socket
Power Switch
2

3. Safety and Regulatory Information
Note:
Read all instructions carefully before use.
The following basic precautions are needed when using an electrical product:
Caution:
Failure to read and observe all precautions could result in
personal injury or equipment damage.
Product cautions:
1. To avoid electrical shock: Keep the unit away from water
• Do not immerse the mains lead or the unit in liquid
• Do not use while bathing
• Do not reach for a unit that has fallen into water
– immediately unplug the unit.
2. Never operate the unit if:
• it has any damaged parts (including mains lead),
• it has been dropped or submersed in water.
3. The unit should not be used where flammable gas, oxygen or aerosol
spray products are being used.
4. Disconnect the unit from the electrical outlet before cleaning, filling and
after each use.
5. Do not use other attachments unless recommended by manufacturer.
Operating cautions:
1. Connect this product to an appropriate voltage outlet for your model.
2. Do not run this product unattended.
3. Never operate if this unit has a damaged lead or plug, if it has been
dropped into water.
4. If any abnormality occurs, discontinue use immediately until the unit has
been examined and repaired.
5. Always unplug the product immediately after use.
6. Never block the air openings of the main unit or place it where the air
openings may be obstructed.
Storage cautions:
1. Do not store the unit under direct sunlight, high temperature or humidity.
2. Keep the unit out of reach of small children.
3. Always keep the unit unplugged while not in use.
Cleaning cautions:
1. Do not immerse the unit in water. It may damage the unit.
2. Disconnect the unit from the electrical outlet before cleaning.
3. Clean all necessary parts after each use as instructed in this handbook.
No modification of this equipment is allowed
3
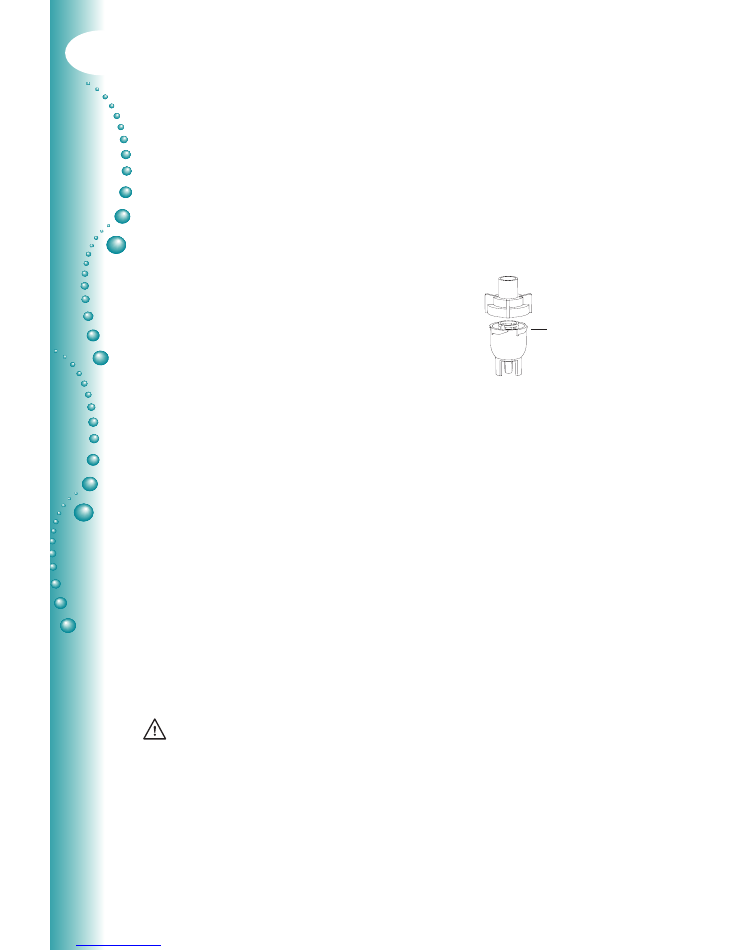
Place cone
inside and
fill with
medication
4
4. Operating Your Compressor Nebuliser
Note:
Prior to initial operation, the nebuliser should be thoroughly cleaned.
1. Place your Compressor Nebuliser on a flat and stable surface. Be sure that
you can easily reach the controls when you are seated.
2. Open the top cover.
Important:
Prior to initial operation, the nebuliser should be thoroughly
cleaned referring to ‘Cleaning’, section 7, page 6 in the handbook.
3. Gently twist the top part of the nebuliser counter-clockwise to disassemble
the nebuliser.
4. Fill the bottom section of the nebuliser with the
medication prescribed by your physician. Be
sure that the cone is put inside the bottom section.
5. Gently twist the top part clockwise to re-assemble
the nebuliser. Be sure the two sections fit well.
6. Attach one end of air tube to the base of the nebuliser.
7. Attach the other end of air tube to the air tube connector located on the
front of the compressor.
8. Attach mouthpiece or mask per your choice to the top section of nebuliser.
9. Plug the mains lead into an appropriate electrical outlet. Make sure at this
stage, the power switch is at “OFF” status.
10. Press power switch to begin your prescribed treatment.
Important:
The compressor motor has a thermal protector which will shut the unit off
before the unit is overheated.
When the thermal protector shuts the unit off, please:
a. Switch the unit off.
b. Unplug the unit from the electrical outlet.
c. Wait 30 minutes for the motor to cool down before another treatment.
Make sure the air openings are not obstructed.
11. When treatment is finished, switch the unit off and unplug it from the
electrical outlet.
Please Note:
The device should be positioned so that the AC adaptor and the mains plug are
easily accessible to disconnect the device.

5. Notes on Nebuliser Care
1. Replacing your consumables
It is recommended that the following disposable items should be changed
as follows:
Nebuliser chamber
Air tube
Face mask
Inlet filter
Mouthpiece
2. Spares and Accessories
The following are available from your supplier:
Inlet filter (Pack of 5)
Adult Year Pack:
Contents: 4 nebuliser cups, 4 air tubes, 4 adult face masks,
5 inlet filters.
Child Year Pack :
As above but with a child’s face mask.
3. Optional Accessories:
Travel bag
6.
Replacement Parts & Optional Accessories
Replacement parts are available through your local retailer or they can be
ordered directly from Clement Clarke International Limited.
5
change every 3 - 4 months
change every 30 days or
when it turns grey
change every 6 - 12 months
}
}
}
6
7. Cleaning
It is recommended that the nebuliser, driveline (make sure the driveline is dry
before re-use), mouthpiece and/or mask be thoroughly cleaned with warm
water after each use and cleaned with a mild detergent after the last treatment
of the day. If your physician or respiratory therapist specifies a different cleaning
procedure, follow their instructions.
Caution:
Before cleaning, make sure the mains lead is disconnected from the mains
outlet and the unit.
Rinsing (after each treatment)
1. Disconnect the nebuliser cup, driveline, mouthpiece and/or mask.
2. Gently twist the nebuliser cup to open it.
3. Rinse the nebuliser cup, mouthpiece and/or mask with water.
4. Dry them with a clean soft towel or let them air dry.
5. Reassemble the nebuliser cup when completely dry and put the parts in a
dry,
sealed container.
Cleaning your accessories:
This procedure does not sterilise the nebuliser cup. Therefore if the patient has
an infectious disease, the entire nebuliser kit should be discarded.
1. After use, disassemble and discard any residual drug.
2. Wash the nebuliser parts in warm water with a little detergent.
3. Rinse in clean warm water and allow to dry completely before re-assembly.
4. Run empty for a few moments before next use.
5. Store in a dry, clean place.
Cleaning the compressor:
1. Wipe daily with a damp cloth.
2. Do not use any powdered cleaners or soap pads, which may damage
the finish.
Filter change
1. Do not use cotton or any other materials. Do not wash or clean the filter.
Only use filters recommended by your manufacturer. Do not operate without
a filter.
2. Change the filter every 30 days or when the filter turns grey.
3. Changing procedure:
a. Remove the filter cover
b. Replace the used filter with a new one.
c. Replace the filter cover.

8. Technical Specifications
Power
AC 230V, 50Hz
Current
180VA
Medication bowl capacity
5ml (cc)
Particle Size
0.5 to 10 μ m
MMAD
4 μ m
Sound Level
Approx. 60 dBA (1m away)
Average Nebulisation Rate
≥ 0.2ml/min
Compressor Pressure Range
29 to 43 Psi (200 to 300 kPa / 2 to 3 bar)
Operating Pressure Range
10 to 16 Psi (70 to 110 kPa / 0.7 to 1.1 bar)
Litre Flow Range
4 ~ 8 L/m
Operating Temperature Range 10°C to 40°C (50°F to 104°F)
Operating Humidity Range
10 to 95% RH
Storage Temperature Range
-25°C to 70°C (-13°F to 158°F)
Storage Humidity Range
10 to 95% RH
Dimension (L x W x H)
175 x 112 x 140 mm (6.88” x 4.41” x 5.5”)
Weight
1.38 kg (3.01 lbs)
Standard Accessories
Mains Lead, Neb Cup, Driveline, Mouthpiece,
Filters (5pcs), Face Mask (Adult & Child)
Protection against electric shock:
- Class II Equipment
- Type BF applied parts
Degree of safety in the presence of flammable anaesthetics or oxygen:
- No AP/APG (Not suitable for use in the presence of flammable
anaesthetics or oxygen)
Mode of operation:
– continuous.
7

8
9. Guarantee
This guarantee is offered to you as an extra benefit and does not affect your
statutory rights.
CCI products are carefully designed, manufactured and inspected.
CCI undertake to replace or repair any part found to be defective in material or
workmanship subject to the following terms and conditions below:
The compressor and motor are guaranteed for 2 years from date of purchase,
on condition that:
•
it is cleaned and maintained in accordance with the instructions.
•
any repair is undertaken by CCI approved engineers/agents.
Please note:
This device does not require a service under its guaranteed life.
This guarantee does not apply to:
•
The disposable nebuliser cup, driveline, face mask, mouthpiece
and filters.
•
Defects arising from misuse, negligence, improper maintentance,
accident, damage in shipment or unauthorised modification or service.
Compensation
Defective parts only will be replaced.
Terms and conditions apply
10. Product Contents
1 x Compressor Nebuliser Unit
1 x Instructions
1 x Neb Cup
1 x Mouthpiece
1 x Adult Face Mask
1 x Child Face Mask
1 x Driveline
5 x Air Filter

11. Symbol Explanations
Consult ‘Instructions for Use’
Caution
Manufacturer
Class II Equipment
Type BF applied parts
This product complies with the essential requirements of the medical
devices directive (93/42/EEC). Compliance has been verified by UK
notified body per 0120 (SGS United Kingdom Ltd).
Do not dispose of with household refuse
12. EMC Declaration
With the increased number of electronic devices such as PCs and mobiles,
medical devices in use may be susceptible to electromagnetic interference from
these devices. Which may result in incorrect operation of the medical device
and create a potentially unsafe situation. Medical devices should also not
interfere with other devices.
In order to regulate the requirements for EMC with the aim to prevent unsafe
product situations, the EN60601-1-2 standard has been implemented. This
standard defines the levels of immunity to electromagnetic interferences as well
as maximum levels of electromagnetic emissions for medical devices.
Medical devices manufactured by Clement Clarke International Ltd conform to
this EN60601-1-2 standard for both immunity and emissions.
Nevertheless, special precautions need to be observed:
•
The use of accessories and cables other than those specified by
Clement Clarke Int. Ltd, with the exception of cables sold by Clement Clarke
International Ltd. as replacement parts for internal components, may result in
increased emission or decreased immunity of the device.
•
The medical devices should not be used adjacent to or stacked with other
equipment. If adjacent or stacked use is unavoidable, the medical device
should be seen to operate normally as it should in this situation.
Further guidance regarding the EMC environment (in accordance with
EN60601-1-2) in which the device should be used is available at
www.clement-clarke.com/ medicalquality
9

Clement Clarke International Ltd
Edinburgh Way Harlow
Essex CM20 2TT UK
Tele: +44 (0)1279 414969
Fax: +44 (0)1279 456300
email: resp@clement-clarke.com
Part no: 3605911
Issue no: 4 01/13